LungPrint® by VIDA
LungPrint Clinical Solutions
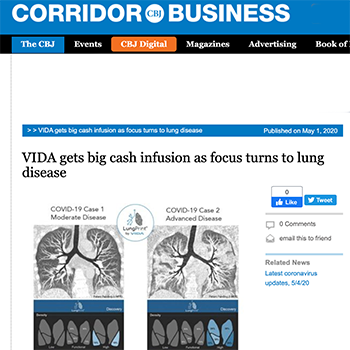
LungPrint® is an AI-powered suite of products and services used to uniquely profile and manage the patient with, or at-risk of, respiratory diseases. LungPrint Clinical Solutions provide clinically validated lung imaging analysis to healthcare providers to obtain a more precise, personalized profile of pulmonary health. LungPrint is FDA cleared, CE-marked, Health Canada licensed and TGA registered for clinical use in the US, European Economic Area, Canada and Australia.
LungPrint Clinical Trial Services
For pulmonary device or pharmaceutical vendors conducting clinical trials, VIDA operates an expert core lab quality controlled by an ISO-13485 certified quality system. With over 30 validated imaging biomarkers, LungPrint Clinical Trial Services offers the highest quality quantitative CT (QCT) analysis for all major respiratory diseases, including COPD (small airway, emphysema), asthma, cancer, interstitial lung disease (ILD) and others.
Our Mission
VIDA is transforming lung care through intelligence, using imaging-based AI to uniquely profile and manage the patient with or at-risk of respiratory diseases. VIDA’s LungPrint® solution aims to provide greater precision and personalization across a range of lung diseases including lung cancer, obstructive airway diseases like emphysema and asthma, and interstitial lung disease. VIDA’s software and services are available for clinical use in the USA, EU, Canada and Australia.